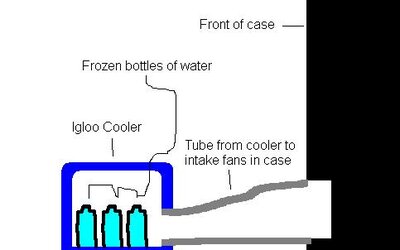
Would the setup in the picture cause condensation inside the computer? There would probably be 6-8 bottles of frozen water inside the cooler as opposed to the three in the picture. There would be a hole cut in the cooler to allow an insulated tube to connect the cooler to the intake fans in the computer case. Tell me what you think.
-
Welcome to Overclockers Forums! Join us to reply in threads, receive reduced ads, and to customize your site experience!
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Cooler idea
- Thread starter Radical
- Start date
f00t said:no. the temp change woul not be drastic enough and the water would soon heat up
Wrong. It depends. What are your outside air conditions? Blowing 35-40 degree air into a 100+ degree case will sure as hell cause condensation if there is ANY humidity in the air.
rhino56 said:the amount of cfm that would be passing thru the bottles would require a huge cooler with lots of bottles because your just radiating the cold.
What??
- Thread Starter
- #5
The outside air conditions would probably be about 28 or 29C, and the case temp would probably be around 29C.
Also, I was thinking of mounting a fan on the end of the tube that's inside the Igloo cooler. The fan would blow air from the cooler toward the intake fans on the case.
Also, I was thinking of mounting a fan on the end of the tube that's inside the Igloo cooler. The fan would blow air from the cooler toward the intake fans on the case.
- Joined
- Jun 13, 2003
- Location
- Albemarle, NC, USA!
try it, tell us when your computer blows.
Very inventive idea, but not very practicle or worth while IMO. You'd see more of a temp drop SAFELY if you just cut your fan grills, cleaned up your wiring, and found out which fan config works best for your case.
Right now mine is 5x intake, 2x exhaust, maybe you could try something like that.
Find the warm spots in your case and get rid of them, the best thing I can tell you.
Very inventive idea, but not very practicle or worth while IMO. You'd see more of a temp drop SAFELY if you just cut your fan grills, cleaned up your wiring, and found out which fan config works best for your case.
Right now mine is 5x intake, 2x exhaust, maybe you could try something like that.
Find the warm spots in your case and get rid of them, the best thing I can tell you.
- Joined
- Dec 7, 2003
- Location
- Portland, OR usa
Ah no you'll get condensation where warm air drops in temperature - that is, this setup will have lots of condensation around the *bottles* not in the case (where cool air is warming up.)
It should work fairly well for a few hours at least. 10 lbs of water = 4480 grams of 0 C water , that's a lot of joules it can absorb.
4 joules per gram per degree C for water, right? That's 4 *4480 * 20C = 360,000
25 cu ft of air per minute, lowered by 5C, = 31.5 grams per cu ft:
1 Joule to raise 1 g air 1C. 5 x 31.5 grams = 157 joules per minute.
360,000 joules = 2300 minutes or 38 hrs of lowering 25 CFM air 5C.
And that doesn't count the joules used in converting 0C ice to 0C water (lots).
Still going to have to replace bottles every day or two.
the wesson
(edit: forgot total temp rise of 20C, sorry)
It should work fairly well for a few hours at least. 10 lbs of water = 4480 grams of 0 C water , that's a lot of joules it can absorb.
4 joules per gram per degree C for water, right? That's 4 *4480 * 20C = 360,000
25 cu ft of air per minute, lowered by 5C, = 31.5 grams per cu ft:
1 Joule to raise 1 g air 1C. 5 x 31.5 grams = 157 joules per minute.
360,000 joules = 2300 minutes or 38 hrs of lowering 25 CFM air 5C.
And that doesn't count the joules used in converting 0C ice to 0C water (lots).
Still going to have to replace bottles every day or two.
the wesson
(edit: forgot total temp rise of 20C, sorry)
Last edited:
- Joined
- Jun 10, 2003
Yeah, replacing the bottles will be a hassle. Pretty much, every morning I'd put 10 fresh bottles in and put those 10 back in the fridge.
ocforums123
Disabled
- Joined
- Apr 24, 2004
i thought of that same idea only i thought of using Compressed air cans... there super cold. but i was liek nah screw this...
- Joined
- Dec 7, 2003
- Location
- Portland, OR usa
Radical said:Wow, thanks The Wesson. That really puts this idea into perspective. Hopefully I can go and get some bottled water or something today and have this thing running by next weekend.
Much obliged ...
I suppose you'll want to make sure that the air duct doesn't get condensation on it so that condensation could trickle along it and then run into the computer either.
But I see from your picture that the duct is running UP anyhow.
Also you could insulate it.
Overall I wouldn't expect condensation on the outside of the duct to be a big problem. If it is, just increase airflow through the cooler so that output air isn't as cold <grin>
the wesson
Similar threads
- Replies
- 8
- Views
- 218
- Replies
- 4
- Views
- 2K